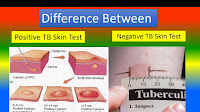
As we move quickly through the new vaccine and await the results of vaccinations, more issues are being found. One concern is the COVID vaccine could trigger a positive TB test result. As of this writing, I have been advised that at least one facility administered the vaccine and, within a few days, completed their annual TB skin test only to have almost everyone test positive.
A quick search on the Internet revealed that the CDC is aware of the issue. There are already several articles addressing the problem as well (example: Article).
A nurse for the facility where the TB test was completed and showed positive after the vaccine recommends updating the TB test first. Most facilities such as ICF programs obtain an annual TB test. If your facility is scheduling a COVID Vaccine, it might be advisable to update all TB tests for the year first. To get specific recommendations or instructions, discuss this with the facility nurse or medical advisor.